As featured in Waterline Winter 2022-23
From the Archive: This article first appeared in waterline Winter 2019-2020
Effect of sample pH on the identification of an NRB in Closed Hot and Cold Water Systems
Pamela Simpson (PhD), Whitewater Technologies Ltd; Richard Jones (MSc), Cheshire Scientific Ltd.
Introduction
A closed re-circulating pipework system is one which, as the name implies, is closed i.e. the water in them is not exposed to the atmosphere and is not significantly depleted due to evaporation or draw-off. The water is permanently enclosed and typically spends all of its time being heated, cooled and re-circulated in the process of delivering heating or cooling.
The potential problems in closed systems can start during construction. In large buildings, heating and cooling circuits can include pipes that are over a metre in diameter. In an ideal world, these pipes would be installed in a clean, debris-free condition but in practice, this is not always the case.
Pre-commission cleaning of closed circuit pipework systems and the subsequent monitoring of water quality are essential in any building. The implications of getting these wrong can be catastrophic. The resulting problems include disruption to occupants whilst systems are re-cleaned or, in the worst cases, complete closure of buildings whilst entire systems are ripped out and replaced due to early failure.
In theory, once the system is put into operation, the corrosion process should be controllable. If there is no replacement of the water in the system, the oxygen in the water should gradually become depleted thereby stifling the corrosion. Furthermore, corrosion inhibitor chemicals (such as nitrite and molybdate) should be added to further reduce the rate of corrosion. However, these may be depleted in a closed system by developing protective layers on pipes or reacting with any remaining oxygen in the water. Of more concern is the realisation that some inhibitors can provide a food source for certain bacteria, known as NRBs, especially if an appropriate biocide has not been dosed or maintained within the system.
Sources of Bacteria and Associated Problems
All natural sources of water (including mains water) contain many different types of bacteria, some of which may multiply and lead to problems within closed systems if they encounter suitable conditions for growth. Mild steel, stainless steel and copper are thought to be particularly prone to microbial influenced corrosion (MIC). For MIC to occur, it is necessary for some types of bacterial species to colonise the metal surface. The extracellular material produced by rapidly multiplying aerobic bacteria species e.g. Pseudomonas spp develops into a biofilm (i.e. slime) which produces both aerobic and anaerobic zones. The low oxygen levels within the system and under the biofilm enable anaerobic bacteria, e.g. sulphate reducing bacteria (SRB), to multiply and these species can be associated with localised pitting corrosion.
Another further species often associated with a biofilm is the NRB, Pseudomonas denitrificans. This species can enter closed systems via fill or top-up water during commissioning and will multiply if given an adequate food source such as a nitrite corrosion inhibitor. Loss of this corrosion inhibitor may contribute to a decrease in metal surface protection, but also an increase in corrosive ammonia and nitrogen gases within the water system. It is therefore essential to be able to identify the presence of NRB within a closed system to prevent pipe surfaces being left vulnerable to corrosion.
Microbial Water Analyses of Closed Systems and Pitfalls when looking at NRB
Often water analyses can provide useful information about the state of a closed system. However, on occasion, the results obtained for bacteria, in particular NRB, can be misleading and the detection of NRB should be correlated to the water chemistry results. Table 1 illustrates how the detection of NRB is considered unlikely on 4/12/17 as neither TVC nor Pseudomonas spp were detected suggesting that the water was of acceptable quality microbiologically. However NRB were recorded as “heavy growth” and the nitrite levels were low suggesting that NRB had consumed the inhibitor. But is this the case?
As Pseudomonas spp. are one of many denitrifying bacteria, their presence (certainly if present as “heavy growth”) would be detected in the TVC analysis and possibly also in the Pseudomonas spp. analysis. Although NRB have specific nutrient requirements for growth, some NRB species (other than Pseudomonas spp) may grow on both NRB Sig Nitrite Media and TVC Yeast Extract Agar but may be absent from a Pseudomonas Agar plate. Thus NRB species would always be detected on either TVC or Pseudomonas growth media if “heavy growth” has been recorded in the NRB test.
Table 1 on the next page illustrates the point that the NRB recorded (as heavy growth) may be a false positive, especially in light that no bacteria are recorded for TVC and Pseudomonas spp.. Low levels of nitrite inhibitor, in this case, were more likely attributed to the inhibitor being absent or at a low concentration.
In this example, the system water pH was 9.9 (high for a closed system, usually in the range 7 – 9.5) and it was considered that the high pH may be contributing to the false positive of the NRB test. Laboratory techniques often use a Sig Nitrite Test. This utilises the production of ammonia and nitrogen gas by NRB to detect their presence in aqueous solutions. A positive reaction for this test is indicated by a pink colouration of the media as the production of ammonia by NRB increases the media’s pH and/or the production of bubbles in the media by nitrogen or other gas production.
Water samples from closed systems often have a high alkalinity and have been shown to produce an instant pink colouration in the Sig Nitrite Media test. The colouration is intensified the longer the samples are left. If looking for a colour change as part of the laboratory protocol, these samples would be recorded as having “heavy growth” even though NRB are absent, leading to a false positive result and possibly unnecessary re-dosing of a system with biocide.
Table 1: Typical water analysis for a LTHW closed system.
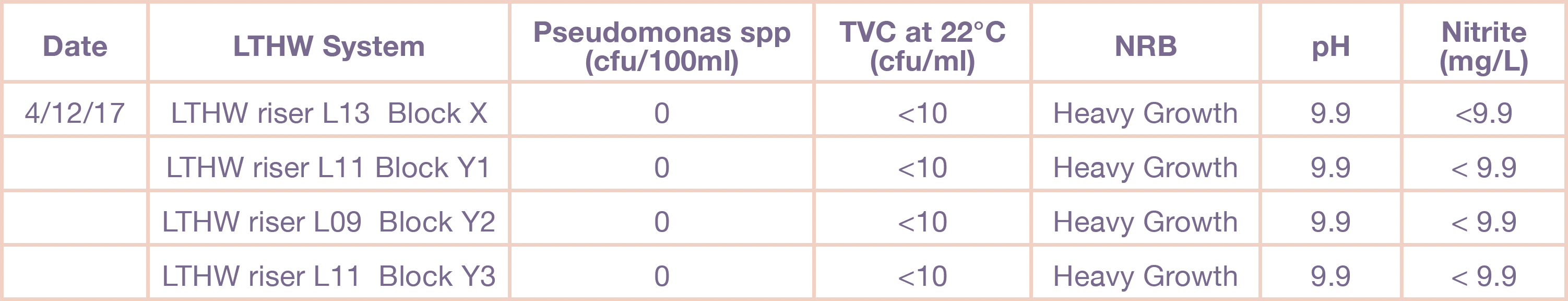
Controlled laboratory Investigation to Correlate pH with Colour ChangeA laboratory investigation was carried out to identify if pH could contribute to a false positive as observed by a colour change. Figure 1 below illustrated how sterile water samples influenced the colour change when pH was altered.
Figure 1: Effect of pH on the colour change in sig Nitrite Test Media after 48 hours and 5 days respectively.
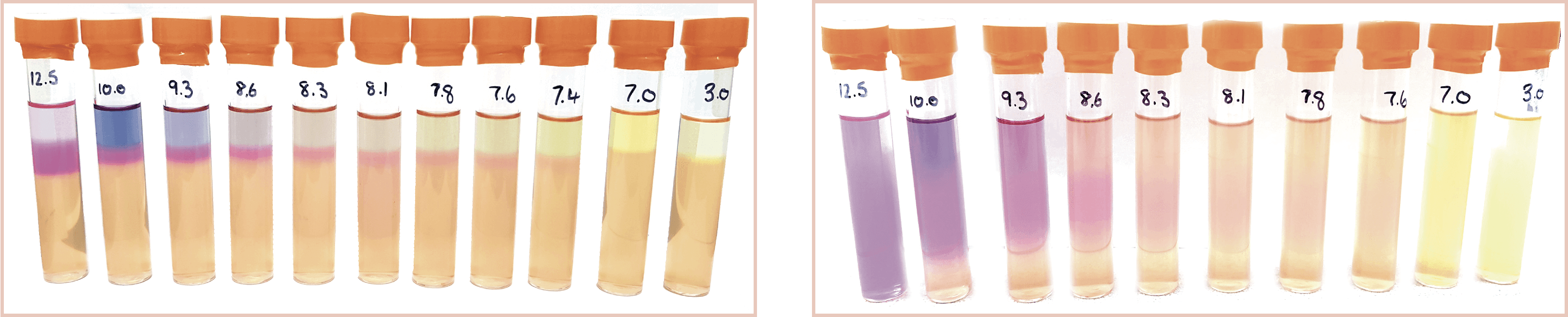
These tests confirmed that the effects of sample pH on the interpretation of Sig Nitrite Test results were deemed negligible at less than pH 7.4, but significantly altered the colouration above this pH which, for closed systems, would give a false positive result each time.
One other criteria of this test was to look for bubbles of ammonia gas produced by the growth of NRB bacteria within the media. However further studies using water samples inoculated with Pseudomonas denitrificans showed that bubbles were not produced by this species at any pH tested. A more intense pink colouration formed however, and it would be advised to set up a sterile blank of the same pH value for samples above pH 7.4. This would then be incubated alongside the sample and used as a comparison to differentiate the pink colouration caused by ammonia production from the pink colouration caused by sample pH. As discussed, P. denitrificans may not produce copious gas bubbles in the Sig Nitrite media and this may also be the case for other NRB species, thus it is only possible to rely on the pink colouration caused by ammonia production and differentiate this from the pink colouration caused by high sample pH in order to confirm the presence of NRB.
Conclusion
Closed water systems operate at a water pH in excess of 7.5. The methodology used within laboratories to identify NRB species is known as a Sig Nitrite Test. Often water results are reported as having heavy growth of NRB even in the absence of the detection of Pseudomonas species and Total Viable Counts (TVC). Having carried out a study using sterile pH adjusted water, it became apparent that the media used in the Sig Nitrite Test is pH sensitive and a pink colouration occurs in the absence of NRB species at a pH as low as 7.4. Furthermore, the NRB species, Pseudomonas denitrificans, did not produce bubbles of gas which are often used as a second indicator of NRB activity within the test. These species did contribute to a colour change within the test media and at a higher pH (i.e. greater than pH7.4 and characteristic of closed systems) it is advised that a sterile pH adjusted control is included in the test to compare the intensity of the pink colouration. It is hoped that by the inclusion of a sterile blank that false positives will be reduced within the closed water industry and avoid any unnecessary addition of biocides and/or system re-cleans.
About the Authors
Dr Pamela Simpson is a Chartered Fellow of the Society of Biology. She established Whitewater Technologies in 1998, before which she spent over eight years working in the speciality chemicals industry, initially as a Technical and European Director of the Industrial Biocides Division of a major chemicals manufacturing and processing company. She has developed a broad knowledge of the application of microbial control techniques in product preservation and antimicrobial surface protection, process water control, and microbial issues within hot and cold closed systems for both healthcare new-builds and commercial premises. She is also an approved trainer for Legionella awareness courses for water treatment engineers.
Her recent work involved expert work for microbially-influenced corrosion in a range of commercial and healthcare buildings of hot and cold closed systems. She was on the Steering Group for the writing of BSRIA BG50/2013: Water treatment for closed heating and cooling systems.
Richard Jones (MSc) is a skilled and confident microbiology technician at Cheshire Scientific Laboratories, with 8 years experience in bacteriological water analysis using a broad range of traditional culture and molecular biology assays, further expanding his skill set in 2017 by obtaining a MSc Biomedical Science Distinction.












