As featured in Waterline Winter 2023-24
The future for heating in the UK:
Surfactant stabilised formulations for improving energy transfer
Robert Wilson 1, *, Mohammed Yaseen 2, *, Ahmed Etayeb b
*Corresponding Author
Abstract
Next to transport, the heating and cooling1 of our buildings is the largest user of energy in the UK. Around 35% of UK energy is used in this way and 80% to 85%2 of this energy is used to heat water in our heating systems. While the design of the hardware of heating systems has been optimised, nobody has paid much attention to the water heated by boilers and circulated through radiators and fan coils to warm our houses and buildings. Water is an inexpensive, safe material which conducts heat. It is not very efficient as an energy transporter however, because to put it simply, water is not wet.
There is now a group of siliconebased surfactants that can, at low concentration, reduce the surface tension of water to about 20 mN/m, and the contact angle between water and a solid (steel in a radiator) to 0o. A zero-contact angle means complete wettability. This type of surfactant is known as a super spreader. It has been demonstrated that complete wettability caused by a zero-degree contact angle means that 3 times as much water is present on a surface than can be achieved by using a standard Alkyl Poly Glycoside (APG) based surfactant.
Applying this technology to a building heating system means that a small amount of this superspreader, dosed into the water in a heating system will ensure that spaces heat faster, and boilers operate less, saving an estimated 20% of the energy previously used. The superspreader has been commercialised and this product will give a payback of less than one year – based on current energy costs. If this technology were applied to the water of every water based heating and chilled water system in the UK the yearly energy saving would equate to the annual output of 2 Drax B3 power stations.
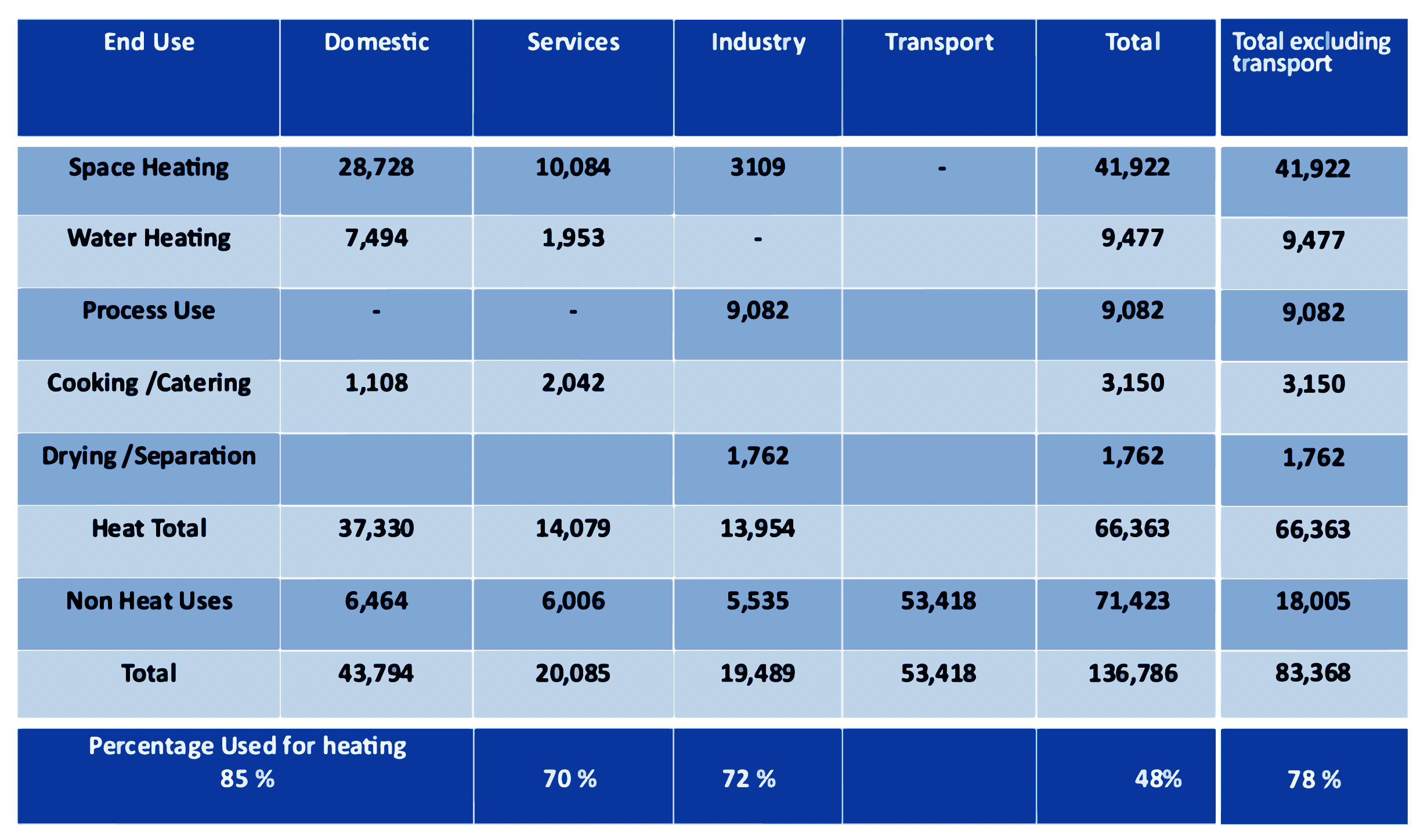
Table 1: How energy is used in the United Kingdom, (UK government figures 2013)4
Introduction
Almost 40% of all the energy used in the UK is used to heat our homes and places of work. Nearly all this energy (85%) is used to heat water. This water is heated in boilers, by heat pumps or other means to temperatures up to 80oC and circulated through radiators or fan coils to heat individual spaces. The efficiency of boilers, heat pumps etc and radiator/fan coil design has been optimised to the extent that further improvements would not improve fuel savings.
Until recently, nobody has paid much attention to the water that flows in the closed heating loop, believing that water is the ideal cheap, safe, and thermally stable heat transfer medium. Its efficiency for transferring heat from the boiler to the air in a room has been taken for granted. Water, however, is a poor conducting medium, because it is not very wet and makes poor contact with the metal surface of a radiator or fan coil.
While Wilson postulated that the surface tension of water is related to its thermal conductance 10 years ago, it is only in the past 2-3 years that others have published papers indicating agreement. The surface tension of water is related to the angle which a drop of water makes with a surface. This is known as the contact angle. The addition of a low concentration of a surfactant will significantly reduce the surface tension of water and also reduce the contact angle, increasing the water contact with the surface, and make water wetter. This is important because the metal surface of a radiator is full of tiny micro and nano crevices that water cannot enter. This is the basis of commercial products already on the market.
At The University of the West of Scotland, Dr Yaseen’s research lab has shown that water containing a superspreader surfactant will heat a room much faster than water itself and faster than water containing other surfactants. More importantly water containing a superspreader surfactant with nanomaterial formulation will heat a room faster than water containing metal or graphene nanoparticles on its own. Nanoparticles make the water more thermally conductive, with surfactants being important because they make the surface wetter.












